You are here
Fungi
Ustilaginomycotina
EOL Text
The class Ustilaginomycetes comprises more than 1400 species of basidiomycetous plant parasites, which are distributed in approximately 70 genera. They occur throughout the world, although many species are restricted to tropical, temperate or arctic regions. Some species of Ustilago and Tilletia, e.g. the barley, wheat or maize smut fungi, are well known because they are of economic importance. For example, from 1983 to 1988 the barley smut fungi reduced annual yields by 0.7% to 1.6% in the prairie provinces in central Canada, causing average annual losses of about U.S. $8,000,000 (Thomas 1989). Tilletia contraversa is important in the international wheat trade (Trione 1982), and 2-5% of the plants in a corn field are generally infected by Ustilago maydis, while up to 80% of a field can be infected if conditions are good for the smut fungus. On the other hand, the galls of U. maydis are considered a delicacy in the Mesoamerican tradition. They are known in Mexico as "Huitlacoche" and in the U.S.A. as "maize mushroom", "Mexican truffles" or "caviar azteca" (Valverde et al. 1995).
| License | http://creativecommons.org/licenses/by/3.0/ |
| Rights holder/Author | Robert Bauer, Dominik Begerow, Franz Oberwinkler, Tree of Life web project |
| Source | http://tolweb.org/Ustilaginomycotina/20530 |
Like the terms agaric, polypore, lichen etc. the term smut fungus circumscribes the organization and life strategy of a fungus, but it is not a taxonomic term. Fungi that look superficially similar to the teliospore-forming members of the Ustilaginomycetes evolved in different fungal groups, e.g. the Microbotryales in the Uredinomycetes (Bauer et al. 1997) or Schroeteria in the Ascomycota (Nagler et al. 1989).
| License | http://creativecommons.org/licenses/by/3.0/ |
| Rights holder/Author | Robert Bauer, Dominik Begerow, Franz Oberwinkler, Tree of Life web project |
| Source | http://tolweb.org/Ustilaginomycotina/20530 |
Lack of membrane bands or caps at the pores and the presence of local interaction zones without interaction apparatus characterize the Entorrhizomycetidae (Bauer et al. 1997). Entorrhiza is the single genus currently identified of this group.
Presence of enlarged interaction zones characterizes the Ustilaginomycetidae (Bauer et al. 1997). This statistically well-supported subclass (Begerow et al. 1997) comprises 33 teleomorphic (with a known sexual stage) and one anamorphic (without a known sexual stage) genera, e.g. Anthracoidea living on Cyperaceae, Cintractia living on Cyperaceae and Juncaceae, Doassansiopsis living on mono- and dicots, Farysia living on Cyperaceae, Melanotaenium s. str. living on dicots, Mycosyrinx living on Vitaceae, Pseudozyma (anamorphic genus), Sporisorium living on Poaceae, Thecaphora living on dicots, Urocystis living on mono- and dicots or Ustilago s.str. mainly living on Poaceae.
The Exobasidiomycetidae differ from the Ustilaginomycetidae by forming local interaction zones and from the Entorrhizomycetidae by having membrane caps at the pores (Bauer et al. 1997). This subclass contains 35 teleomorphic and two anamorphic genera, e.g. Botryoconis living on Lauraceae, Brachybasidium living on Arecaceae, Coniodictyum living on Rhamnaceae, Doassansia living on mono- and dicots, Entyloma living on dicots, Exobasidium living on dicots, Georgefischeria living on Convolvulaceae, Graphiola living on Arecaceae, Malassezia (anamorphic genus), Microstroma living on Juglandaceae and Fagaceae, Tilletia living on Poaceae, Tilletiaria (only known in laboratory) or Tilletiopsis (anamorphic genus).
| License | http://creativecommons.org/licenses/by/3.0/ |
| Rights holder/Author | Robert Bauer, Dominik Begerow, Franz Oberwinkler, Tree of Life web project |
| Source | http://tolweb.org/Ustilaginomycotina/20530 |
- Plant parasitism
- Cellular interaction with primary interactive vesicles
- Cell wall carbohydrate composition with dominance of glucose and absence of xylose
- 5S rRNA secondary structure of type B
- Septal pores without parenthesomes, but in most cases with distinctive tripartite membrane caps or discs
- Life cycle with a parasitic dikaryophase and a saprobic haplophase
Plant parasitism
In contrast with the Urediniomycetes and Hymenomycetes, the Ustilaginomycetes are ecologically well characterized by their parasitism of vascular plants. Two of the more than 1400 species live on lycophytes, one on ferns, two on conifers, whereas all other Ustilaginomycetes parasitize angiosperms with about 810 species on Poaceae and 170 on Cyperaceae. Interestingly, no species has been reported to parasitize Orchidaceae although this family, with about 20,000 species, is one of the largest groups of the angiosperms. With a few exceptions the teliospore-forming species of the Ustilaginomycetes parasitize nonwoody herbs, whereas those without teliospores prefer woody trees or bushes. However, almost all species sporulate on or in parenchymatic tissues of the hosts. Depending upon the species the sori appear in different organs of the hosts, e.g. in roots, stems, leaves, inflorescences, flowers, anthers, ovaries, seeds etc.
Cellular interaction
An important apomorphy for the Ustilaginomycetes is the presence of zones of host-parasite interaction with fungal deposits resulting from exocytosis of primary interactive vesicles (Bauer et al. 1995a, 1997).
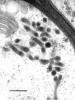

Fig.1. Transmission electron micrograph showing primary interactive vesicles in Exobasidium pachysporum. Scale bar = 0.2 µm. © R. Bauer 1997
Fig. 2. Transmission electron micrograph showing a transfer stage between Mycosyrinx cissi (upper cell) and its host (lower cell). Note the infiltrated host cell wall (between the two arrows) and the deposit at the host cell. Scale bar = 1 µm. © R. Bauer 1997
The contents of these vesicles (Fig. 1) are transferred to the host plasma membrane (Fig. 2). Two major types are recognized. (i) Local interaction zones (Fig. 3): short-term production of primary interactive vesicles per interaction site results in local interaction zones, and (ii) enlarged interaction zones (Fig. 4): continuous production and exocytosis of primary interactive vesicles results in the continuous deposition of fungal material at the whole contact area with the host cell.


Fig. 3. Transmission electron micrograph showing a local interaction zone (arrows) between Exobasidium pachysporum (lower cell) and its host (upper cell). Note the interaction apparatus (arrowheads) and the deposit at the host cell. Scale bar = 0.5 µm. © R. Bauer 1997
Fig. 4. Transmission electron micrograph showing an enlarged interaction zone between Ustacystis waldsteiniae and its host. The haustorium (h) is encased by electron-opaque material. Scale bar = 2 µm. © R. Bauer 1997
Cell wall carbohydrate composition
Prillinger et al. (1993) distinguished between several types of cell wall carbohydrate composition within the Basidiomycota. The Ustilaginomycetes have a distinctive type with dominance of glucose and absence of xylose that separates them from the Urediniomycetes and Hymenomycetes.
5S rRNA
Gottschalk and Blanz (1985) distinguished between a type A and a type B secondary structure of the 5S rRNA. The Ustilaginomycetes share the type B secondary structure with the Hymenomycetes.
Septal pores
In contrast with the Hymenomycetes, the septal pores of the Ustilaginomycetes are without multilayered parenthesomes. In contrast with the Urediniomycetes, in most Ustilaginomycetes the septal pores are enclosed by distinctive, tripartite membrane caps or discs (Bauer et al. 1995b, Bauer et al. 1997, Fig. 5).
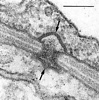
Fig. 5. Transmission electron micrograph showing a typical septal pore apparatus of the Ustilaginomycetes (Entyloma callitrichis) with two membrane caps (arrows). Scale bar = 0.1 µm. © R. Bauer 1997
Life cycle
The Ustilaginomycetes present a rather uniform life cycle with a saprobic haploid phase and a parasitic dikaryophase (e.g. Sampson 1939; Fig. 6). The haploid phase usually commences with the formation of basidiospores after meiosis of the diploid nucleus in the basidium and ends with the conjugation of compatible haploid cells to produce dikaryotic, parasitic mycelia. The dikaryotic phase ends with the production of basidia. In the majority of the Ustilaginomycetes the young basidium becomes a thick-walled teliospore and separates at maturity from the sorus, thus functioning as a dispersal unit. Most of the Ustilaginomycetes are dimorphic, producing a yeast or yeast-like phase in the haploid state. Almost all Ustilaginomycetes multiply mitotically in the saprobic phase, either with yeasts or with ballistoconidia, or with both.
The Ustilaginomycetes share most characteristics of the life cycle with the Microbotryales, which traditionally were considered belonging to the Ustilago-group. However, several independent characters show that the microbotryaceous species of the genera Aurantiosporium, Fulvisporium, Liroa, Microbotryum, Sphacelotheca, Ustilentyloma and Zundeliomyces are actually Urediniomycetes (Gottschalk and Blanz 1985, Prillinger et al. 1993, Swann and Taylor 1993, Bauer et al. 1997).
| License | http://creativecommons.org/licenses/by/3.0/ |
| Rights holder/Author | Robert Bauer, Dominik Begerow, Franz Oberwinkler, Tree of Life web project |
| Source | http://tolweb.org/Ustilaginomycotina/20530 |
Note: The classification of Ustilaginomycotina is being revised. The tree at the top of this page reflects the current classification, whereas the following tree and the text on this page reflect a prior classification.

Since Tulasne and Tulasne (1847) the smut fungi are traditionally divided into the phragmobasidiate Ustilaginaceae and the holobasidiate Tilletiaceae, which are sometimes treated as separate orders (Kreisel 1969, Oberwinkler 1987). Difficulties in their classification have been discussed, e.g. by Durán (1973) and Vánky (1987), neither of whom listed higher taxa in this group. Based predominantly on host-parasite interactions and septal pore apparatus, a radical change in the systematics of smut fungi has been proposed by Bauer et al. (1997). As a result, on the one hand the Microbotryales were excluded from the Ustilaginomycetes and on the other the Exobasidiales s. l., Graphiolales and Microstromatales were included in this group. Cellular interaction (Bauer et al. 1997) and cell wall carbohydrate composition (Prillinger et al. 1993) indicate that the class Ustilaginomycetes in this composition is monophyletic, and phylogenetic analyses of rDNA sequences (Swann and Taylor 1993, Berres et al. 1995, Begerow et al. 1997) are consistent with this hypothesis.
Ultrastructural and rDNA sequence analyses provide evidence for the existence of three major groups within the Ustilaginomycetes (Bauer et al. 1997, Begerow et al. 1997). The basal dichotomy is between the Entorrhizomycetidae and the branch uniting the Ustilaginomycetidae and Exobasidiomycetidae (Bauer et al. 1997, Begerow et al. 1997). In contrast with the Ustilaginomycetidae and Exobasidiomycetidae, the septal pores of the Entorrhizomycetidae are not enclosed by tripartite membrane caps. The Ustilaginomycetidae form enlarged interaction zones, whereas the Exobasidiomycetidae form local interaction zones.
The Ustilaginomycetes represents the sister group of the Hymenomycetes. Type B secondary structure of the 5S rRNA and glucose as major cell wall carbohydrate component are shared by both classes (Gottschalk and Blanz 1985, Prillinger et al. 1993).
| License | http://creativecommons.org/licenses/by/3.0/ |
| Rights holder/Author | Robert Bauer, Dominik Begerow, Franz Oberwinkler, Tree of Life web project |
| Source | http://tolweb.org/Ustilaginomycotina/20530 |